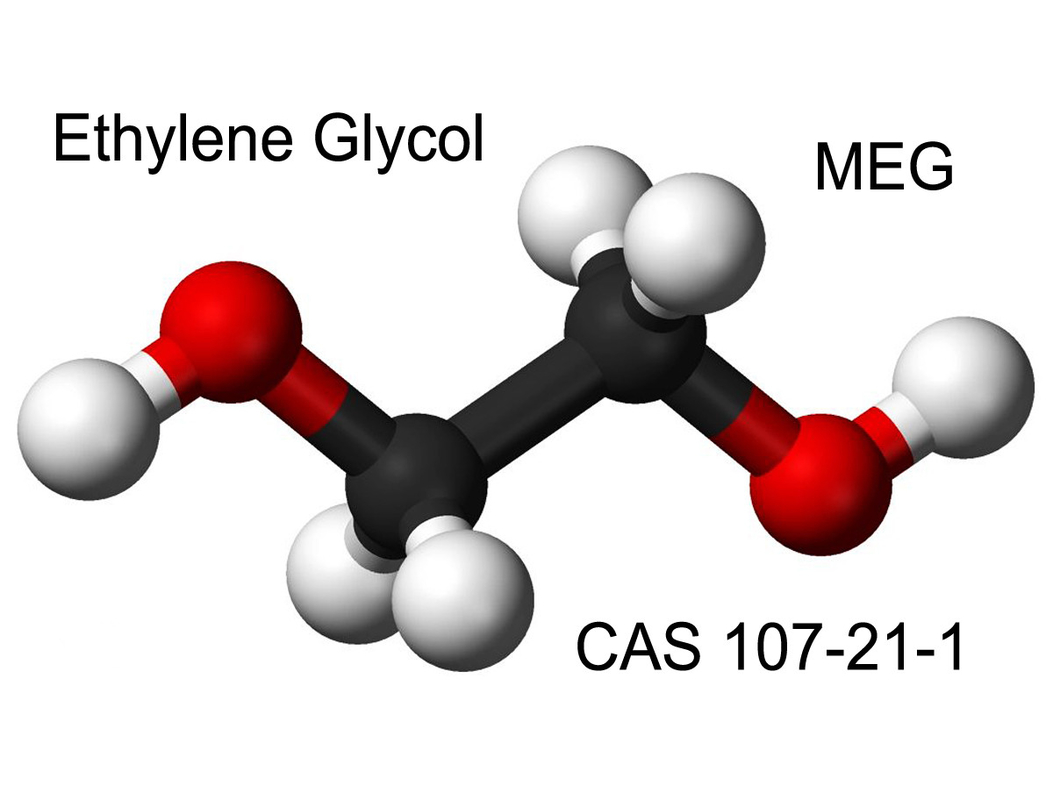
Dihydroxy Alcohol C2H6O2 Mono Ethylene Glycol for Antifreeze Formulations
Quick Details
> Industrial Grade
> Purity 99.0% Min.
> CAS No. 107-21-1
> HS Code 2905310000
> For Chemical, Plastic Industry
> Packing Drum, IBC or ISO tank
Applications
> Coolant
> Anti-freeze
> Hydrate Inhibition
> Dehydrating Agent
> Heat-transfer Agent
> Precursor to Polymers
> Raw Agent for Polyester
> Precursor in Plastic Industry
Our Advantages
> One-stop service
> High quality products
> Professional marketing team
> Stable long-term contract supply
> Short lead time with storage at tank in port
> Served hundreds of customers in 10+ countries
Description
Ethylene glycol is a colorless, practically odorless, low-volatility, low-viscosity, hygroscopic liquid. It is completely miscible with water and many organic liquids. It is completely miscible with water and many organic liquids. This organic compound is highly toxic. It is widely used as an antifreeze and a raw material in the plastic industry.
The hydroxyl groups on glycols undergo the usual alcohol chemistry, giving a wide variety of possible derivatives. Hydroxyls can be converted to aldehydes, alkyl halides, amines, azides, carboxylic acids, ethers, mercaptans, nitrate esters, nitriles, nitrite esters, organic esters, peroxides, phosphate esters and sulfate esters. This chemistry permits ethylene glycol to act as an intermediate in a wide range of reactions. Especially significant is resin formation, including the condensation with dimethyl terephthalate or terephthalic acid resulting in a polyester resin. The reactivity and solubility of ethylene glycol provide the basis for many applications. The widespread use of ethylene glycol as an antifreeze is based on its ability to lower the freezing point when mixed with water. The physical properties of ethylene glycol-water mixtures are therefore extremely important.
Technical indicators (Polyester & Industrial Grade)
| Test Items |
Standard Indicator |
| Polyester Grade |
Industrial Grade |
| Appearance |
Clear Liquid |
Clear Liquid |
| Ethylene Glycol ω/% >= |
99.9 |
99.0 |
| Diethylene Glycol ω/% <= |
0.050 |
0.600 |
| Chroma (Platinum-Cobalt) <= |
5 |
10 |
| Density (20°C) g/cm3 |
1.1128~1.1138 |
1.1125~1.1140 |
| Boiling Point (0°C) |
196.0 |
195.0 |
| Water ω/% <= |
0.08 |
0.20 |
| Acetic Acid (mg/kg) <= |
10 |
30 |
| Iron (Fe) (mg/kg) <= |
0.1 |
5.0 |
| Impurities (mg/kg) <= |
10 |
20 |
| Formaldehyde (mg/kg) <= |
8.0 |
/ |
| Cl- (mg/kg) <= |
0.5 |
/ |

Mono Ethylene Glycol Factory

Mono Ethylene Glycol Loading
FAQ for Ethylene Glycol
1. What is ethylene glycol used for?
Ethylene glycol is a chemical that is widely used in many commercial and industrial applications including antifreeze and refrigerants. Ethylene glycol helps avoid the freezing of your car’s engine in winter, and serves as a coolant to minimize overheating in summer.
2. Is ethylene glycol an alcohol?
Ethylene glycol, also known as ethane-1,2-diol, is the simplest member of the organic compound family glycol. A glycol is an alcohol on adjacent carbon atoms, with two hydroxyl groups. The common name ethylene glycol simply means “ethylene derived glycol.”
3. What is the common name of ethylene glycol?
Ethylene glycol (name IUPAC: ethane-1,2-diol) is an organic formulated compound (CH2OH)2. It is used mainly for two reasons, as a raw material in the manufacture of polyester fibers and formulations for antifreeze. This is an odourless, colourless, viscous liquid with a sweet taste.
4. Is ethylene glycol corrosive?
Ethylene glycol is a Corrosion Source. Most corrosion from ethylene glycol is caused by decomposition at higher temperatures of organic acids (such as glycolic acid).
5. Is ethylene glycol polar?
First, ethylene glycol comprises polar O-H groups; they are polar since oxygen is much more electronegative than hydrogen, and thus appears to polarize the pair of electrons in the O-H bond.

 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!